 
Boyle’s law describes the relationship between volume and pressure in a gas at a constant temperature. At a constant temperature, changing the volume occupied by the gas changes the pressure, as does changing the number of gas molecules. Therefore, the pressure is lower in the two-liter container and higher in the one-liter container. In this case, the force exerted by the movement of the gas molecules against the walls of the two-liter container is lower than the force exerted by the gas molecules in the one-liter container. 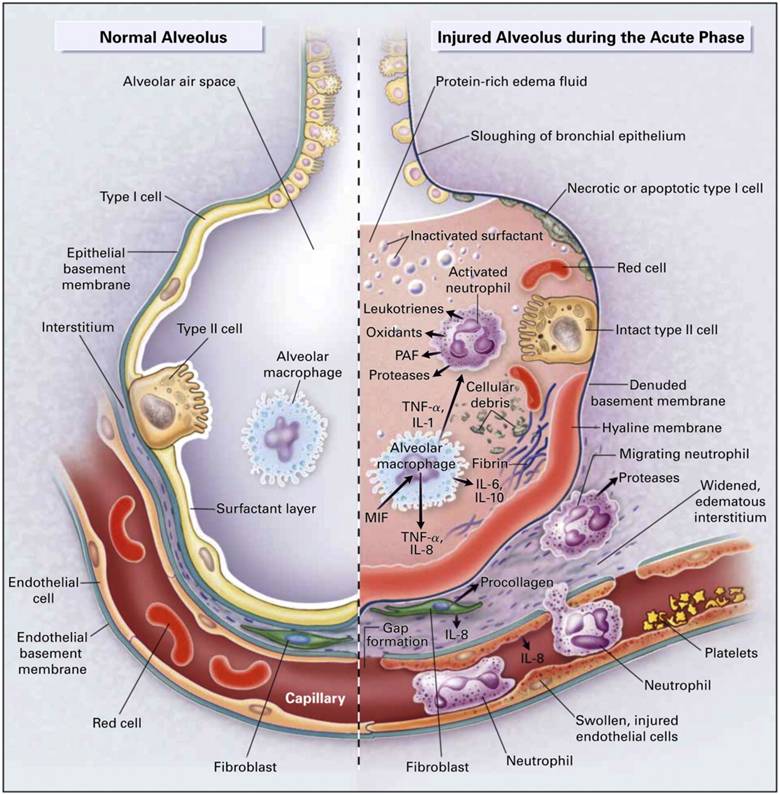
For example, a certain number of gas molecules in a two-liter container has more room than the same number of gas molecules in a one-liter container ( (Figure)). In a gas, pressure is a force created by the movement of gas molecules that are confined. 
Inspiration (or inhalation) and expiration (or exhalation) are dependent on the differences in pressure between the atmosphere and the lungs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
